A sterilization indicator label is a simple visual tool used during the cleaning and sterilization cycle for reusable instruments and packaged materials. It does not replace formal testing methods, but it serves as an immediate, easy-to-read signal that a package or instrument has been exposed to the conditions of a sterilization process. In routine clinical settings, these markers help staff quickly distinguish processed items from those that still require attention, and they contribute to organizational practice around safe handling and inventory control.
These labels work by changing appearance when exposed to certain process parameters. The change is intentionally unambiguous so that a caregiver or technician can confirm at a glance whether a package has been treated. They are applied to instrument sets, trays, pouches, and outer packaging at the point of preparation and remain with the item through the sterilization cycle. After processing, staff are expected to inspect the indicator before storing or introducing the item into a clinical environment.
There are different categories of indicators designed for different purposes. Some are intended for use on the outside of a package to show that the external environment has been exposed to the sterilizing agent. Others are placed inside a package to demonstrate that the interior environment—where instruments sit—was reached by the sterilizing medium. A separate category is used as part of periodic process monitoring and validation; these are used in conjunction with other verification methods and quality records. Each category addresses a particular need within the overall program of sterility assurance.
Choosing the right label for a given use requires attention to how the items will be processed. Factors such as packaging type, sterilization method, and workflow practices influence which label will provide meaningful feedback. The label must be compatible with the sterilization environment so that the indicator responds to the treatment conditions while remaining legible and intact. Labels designed for use under low temperatures differ from those formulated for high-temperature exposure, and labels intended for enclosed packages differ from peel-and-stick versions used on outer wraps.
Proper placement and consistent inspection are essential to getting value from these markers. When applied and read according to established procedures, they reduce the risk of mistakenly using an unprocessed item. Staff should be trained to recognize valid and invalid changes in appearance and to follow an established path for items that show unexpected results. Unexpected results do not necessarily prove contamination but do indicate that an item should be held out of circulation until appropriate checks or corrective steps are completed.
Integration into documentation and traceability practices strengthens the role these labels play in a clinic or hospital. When labels are used alongside batch records, lot identifiers, or tracking systems, they contribute to the chain of custody for instrument sets. This helps facilities maintain clear records for quality reviews, audits, and routine internal checks. A coherent approach to labeling and documentation supports consistent practice across shifts and teams, making it easier to follow procedures and to carry out investigations when questions arise.
In procurement and selection, buyers should look for durable adhesives, clear visual contrast after processing, and labels that tolerate handling during routine storage and transport. Labels that fade with time or degrade with moisture can create ambiguity. It is also important that labeling materials do not contaminate packages or interfere with packaging seals. Where automation or barcode systems are used, labels that are compatible with scanning or with adjacent identification systems can be helpful in maintaining orderly workflows.
Training and competency assessment go hand in hand with the use of these indicators. Staff need practical instruction on where to place labels, how to interpret changes, and what steps to take if a label shows an unexpected result. Simulation and regular review of routine scenarios help build a culture in which indicators are trusted as part of a system rather than seen as a substitute for critical thinking. Competency programs should reinforce the signaling function of the label while emphasizing that it is one element among several in an overall assurance strategy.
A thoughtful approach to storage and handling after sterilization further preserves the usefulness of the visual cue. Packages should be stored under conditions that avoid abrasion, high humidity, or other factors that might alter the label after processing. When labels are used for inventory rotation, placement should allow staff to read the change without opening the package. Consistency in where labels are applied reduces the chance of missed checks and makes visual inventories faster and more reliable.
Communication between teams that handle instruments and those that use them is an important operational consideration. Clear local rules about how long an item is held after processing, how labels are used in mixed inventories, and who is authorized to remove or alter a label will prevent misunderstandings. When changes in practice are introduced, a short period of overlap with supervisory checks helps ensure that the new approach is working as intended.
Selecting the right Sterilization Indicator Label is an important step in maintaining an organized and reliable sterilization workflow. In healthcare facilities, laboratories, and dental clinics, reusable instruments move through multiple stages: collection, cleaning, packaging, processing, storage, and final use. Each stage depends on clear communication among staff members. A visual process indicator supports that communication by showing whether a packaged item has been exposed to the intended sterilization environment.
Content
- 1 Understanding the Purpose of the Label
- 2 Match the Label to the Sterilization Method
- 3 Consider External and Internal Monitoring Needs
- 4 Evaluate Adhesion and Material Stability
- 5 Visual Clarity and Ease of Interpretation
- 6 Practical Evaluation Before Adoption
- 7 Integration Into Quality Practices
- 8 Concluding Perspective
Understanding the Purpose of the Label
Before comparing different products, it is necessary to understand what the label is designed to do. A process indicator does not confirm sterility by itself. Instead, it demonstrates that a package or instrument set has undergone exposure to certain process conditions such as temperature, chemical agent contact, or time-related parameters within a sterilizer.
Because of this limited but important role, the label should be considered part of a broader assurance system. Facilities commonly combine process indicators with internal monitoring, routine equipment checks, and documentation practices. When the label is used correctly, staff can quickly identify whether an item should move forward in the workflow or remain under review.
Match the Label to the Sterilization Method
Different sterilization technologies operate under different environmental conditions. A label that reacts properly in one environment may not function correctly in another.
Common processing methods include:
- High-temperature steam processing
- Low-temperature gas-based processing
- Low-temperature plasma or similar technologies
Each method exposes materials to distinct temperature ranges, humidity levels, and chemical conditions. The label material and indicator ink must respond only when the intended process occurs. If a label reacts during storage or transport, it can create confusion. Conversely, if it does not react during processing, it may fail to provide useful information.
Review the instructions for use provided by the manufacturer and confirm that the label is specifically intended for the processing method in use.
| Sterilization Method | Typical Application Environment | What the Indicator Should Show | Recommended Label Characteristics |
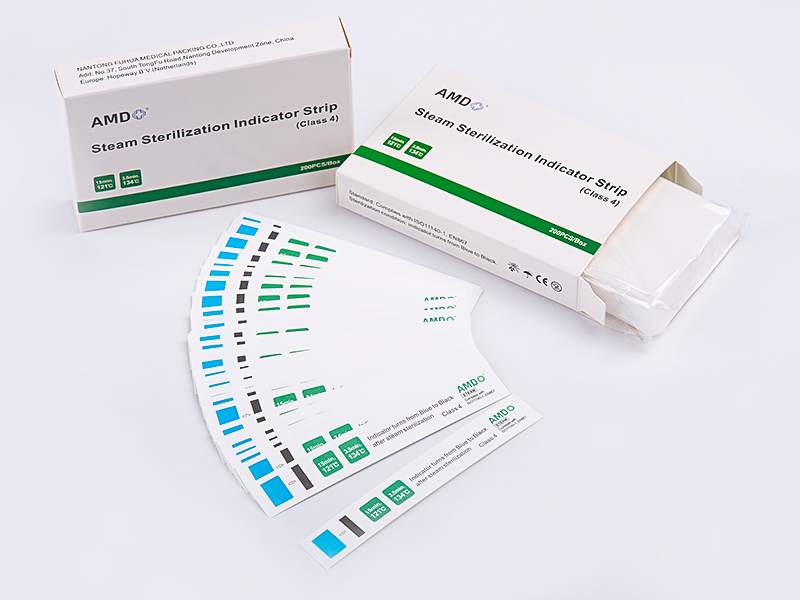
| Steam Processing | Hospitals, surgical centers, dental clinics | Exposure to heat and moisture conditions | Heat-resistant material, stable adhesive, clear visual change after cycle |
| Low Temperature Gas Processing | Heat-sensitive medical devices | Exposure to gas sterilizing conditions | Chemical-resistant ink and packaging compatibility |
| Plasma or Similar Low Temperature Systems | Precision instruments and delicate materials | Exposure to controlled low temperature environment | Non-smearing surface and stable color transition |
Consider External and Internal Monitoring Needs
Facilities often use two forms of monitoring: external and internal. Understanding this difference helps determine which type of label to purchase.
External indicators are attached to the outside of packaging. They help staff distinguish processed packages from unprocessed ones during storage and handling. This is especially helpful when multiple sets of instruments are processed together and then stored in the same area.
Internal indicators are placed inside the package, usually near the instruments. These indicators provide information about whether the sterilizing environment reached the interior of the pack. This is important because packaging materials and loading patterns can influence how the sterilizing medium penetrates the pack.
Some facilities choose to use both types as part of routine practice. When evaluating products, verify whether the label is intended for external placement, internal placement, or both.
Evaluate Adhesion and Material Stability
A suitable label must remain attached throughout the entire processing cycle and storage period. Weak adhesion can cause the label to detach during handling or within the sterilizer. Detached labels create uncertainty and may require reprocessing.
Material stability is equally important. After exposure to heat or chemical agents, the label should remain readable and intact. Staff need to read the visual change easily without opening the package or touching the contents. If the label becomes brittle, smeared, or discolored beyond recognition, it cannot serve its intended function.
Additionally, the adhesive should not interfere with packaging seals. Packaging integrity is part of the sterility assurance approach, so labels must be compatible with wrapping materials such as medical paper, pouches, or nonwoven wraps.
Visual Clarity and Ease of Interpretation
A label is useful only if staff can interpret it consistently. The visual change should be clear and recognizable under routine lighting conditions.
Training programs should include examples of acceptable and unacceptable indicator changes. When selecting a label, consider how easily a new employee could learn to recognize the change after standard instruction.
Facilities sometimes run trial cycles with sample materials to confirm that the visual signal is distinguishable before committing to routine use.
Practical Evaluation Before Adoption
Facilities often evaluate supplies before incorporating them into daily routines. A sample batch can be processed through normal cycles, inspected after cooling, and reviewed after storage. Staff can then confirm whether the label performs consistently under local conditions.
Reviewing written guidance and product specifications is also part of this evaluation. Procurement teams and infection control personnel typically collaborate during the decision process. This helps ensure that the chosen product aligns with established policies and workflow practices.
Integration Into Quality Practices
A labeling system is effective when it complements other monitoring measures. Routine equipment checks, documentation, and staff communication all work together. The indicator provides a quick visual cue, while other verification steps provide broader assurance.
Using the label consistently encourages standardized procedures. When all personnel follow the same steps, packages are handled in predictable ways. This reduces uncertainty and simplifies training for new employees.
Concluding Perspective
Hopeway AMD offers a labeling option intended to assist facilities in identifying processed packages within daily sterilization operations. When incorporated into established procedures, a Sterilization Indicator Label acts as a communication tool rather than a standalone verification method. It helps staff recognize processing status, supports orderly storage, and contributes to documentation practices.
Successful use depends on correct placement, appropriate staff training, and alignment with facility policies. By integrating visual indicators with routine checks and clear procedures, organizations can maintain structured workflows and consistent handling of reusable instruments.

 English
English Français
Français Deutsch
Deutsch Nederlands
Nederlands












 ‘s-Gravenweg 542, 3065SG RotterdamThe Netherlands
‘s-Gravenweg 542, 3065SG RotterdamThe Netherlands
 +31 (0)10 254 28 08
+31 (0)10 254 28 08







