Sterilization is a routine requirement in clinical environments, yet its effective management demands attention to process controls and clear verification methods. For healthcare facilities, dental practices, and scientific laboratories, establishing consistent routines for sterilization monitoring is a central element of risk management and patient safety.
Content
- 1 Why sterilization verification matters
- 2 Types of indicators and where chemical indicators belong
- 3 How indicator tape supports day to day operations
- 4 Correct placement and interpretation
- 5 The chemistry behind the visible change
- 6 Why steam-specific response matters
- 7 Proper placement and reading of indicators
- 8 Integration into clinical workflows
- 9 How the methods differ in principle
- 10 Typical roles in a sterilization program
- 11 Practical application scenarios
Why sterilization verification matters
Sterilization is intended to render reusable instruments and certain disposables free from viable microorganisms before they contact sterile tissues or mucous membranes. Failure to confirm that sterilization cycles have achieved the intended conditions can increase the chance of instrument contamination, and create regulatory exposure. Verifying sterilization is not only a technical step; it is also part of maintaining traceability, meeting inspection expectations, and supporting clinical decision making about instrument use.
Types of indicators and where chemical indicators belong
Verification approaches for sterilization range from routine physical monitoring of equipment parameters to biological testing that confirms microbial inactivation under test conditions. Chemical indicators offer a visual, immediate signal that particular exposure conditions were met during a sterilization cycle. They are commonly applied to instrument packs, trays, or external packaging where a color change makes it straightforward to determine whether items were processed through a steam cycle. While chemical indicators are not a substitute for other forms of validation or periodic biological testing, they provide an accessible method for frontline staff to confirm that an item passed through the sterilization environment.
How indicator tape supports day to day operations
In busy clinical settings, staff need methods that are reliable, easy to interpret, and compatible with existing sterilization logistics. Chemical indicator tape adheres to packaging and labels the pack with a clear visual reference after processing. This reduces uncertainty when sets are staged for storage or immediate use, and it helps prevent inadvertent introduction of unprocessed items into sterile fields. For inventory management, tape that retains legibility after handling enables teams to sort and shelve processed items with reduced risk of confusion. Consistent use of indicators also helps documentation workflows by providing a simple check that can be noted in routine records.
Correct placement and interpretation
The value of a chemical indicator depends on correct use. Indicators should be placed so they are exposed to steam flow within a pack or on the external surface of a wrapped tray where exposure will reflect the internal conditions. After a cycle completes, staff should compare the indicator's appearance against the manufacturer's guidance. A clear and readable change reduces ambiguity when making go or no-go decisions for instrument sets. If an indicator does not show the expected change, the set should be treated as potentially unprocessed and handled according to facility policy.
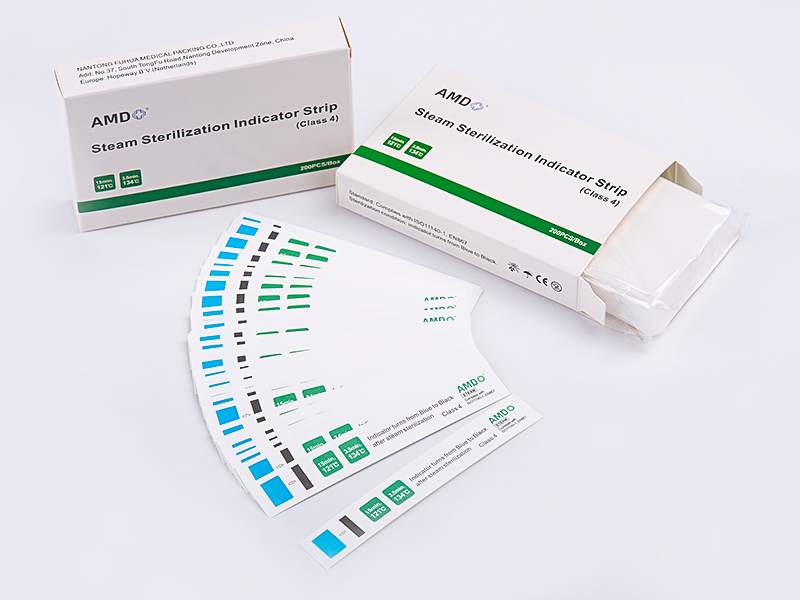
Sterilization is a routine expectation in clinical practice, yet the processes behind it are not always visible to staff who rely on sterile instruments every day. One practical tool that provides a direct visual signal after steam sterilization cycles is the Steam Indicator Strips Tape.
The chemistry behind the visible change
At the heart of a chemical indicator is a reactive formulation that responds to a combination of heat, moisture, and time. These formulations typically include a heat-sensitive dye or dye precursor together with a reactive compound that undergoes a structural change when exposed to saturated steam at the intended sterilization conditions. The change alters the dye's light-absorption properties, producing a different hue that is easy to see by eye.
In a steam environment, the presence of moisture is crucial: steam acts as both a heat transfer medium and a participant in the chemical environment. Under the pressure and temperature conditions present in autoclave cycles, hydrolysis or other steam-facilitated reactions become kinetically favorable and cause the indicator chemistry to progress toward its color state. The indicator design balances sensitivity so that it changes under the intended sterilization conditions but remains stable during storage and handling. Manufacturers produce color references that show the expected outcome; users compare the processed indicator against that reference to determine whether the strip or tape experienced the sterilization environment.
Why steam-specific response matters
Steam sterilization is distinct from dry heat or chemical vapor sterilization because it relies on saturated steam penetration and latent heat to inactivate microorganisms. Indicators formulated for steam exposure are tuned to respond to both the temperature and the presence of steam, reducing the chance of a misleading change from dry heat or mere high temperature without adequate moisture. This specificity helps frontline staff make practical decisions about whether an item has at least passed through a steam cycle.
It is also important that indicators respond visibly and reliably after exposure without requiring specialized equipment to read them. A clear, stable color transformation that remains legible after handling supports routine workflows in busy processing areas where rapid decision making is common.
Proper placement and reading of indicators
To provide a meaningful signal, indicators must be positioned so they experience conditions representative of the load interior. For wrapped instrument trays or pouches, indicators are often placed inside the pack near the area difficult for steam to reach, and an additional indicator may be applied to the external surface to provide a quick visual cue outside the package. For sterilization tape used on outer packaging, the adhesive strip both secures the wrap and provides an external indication that the package has been exposed.
After cycle completion and cooling, staff should compare the indicator's color to the manufacturer's reference. A clearly identifiable change supports routine handling decisions; an absent or ambiguous change should prompt the set to be considered unprocessed until facility procedures determine otherwise. Consistency in placement and interpretation across shifts reduces variability and helps maintain reliable daily practice.
Integration into clinical workflows
In hospitals, dental clinics, and laboratories, indicators are one element of a layered sterilization assurance approach. Typical practice pairs routine physical monitoring of autoclave parameters and scheduled biological verification with the day-to-day use of chemical indicators. In a central processing area, indicators help technicians sort trays, prioritize inventory, and prevent accidental use of unprocessed instruments. In a dental operatory setting, a small indicator inside a pouch provides immediate confirmation when preparing for a patient procedure. In research laboratories, indicator strips assist in separating processed materials from those awaiting sterilization.
For documentation and audit readiness, recording the presence and outcome of indicators on processing logs strengthens traceability. Embedding the check into standard operating procedures and turnover checklists makes the practice routine rather than optional.
Effective sterilization practice relies on multiple verification methods, each answering a different question about the process. Chemical indicators give an immediate visual cue that a package has been exposed to a sterilization environment, while biological indicators provide evidence that the process achieved the conditions required to inactivate resistant microbial forms.
How the methods differ in principle
Chemical indicators are formulated to change appearance when exposed to specific combinations of heat, moisture, and time. They operate through a designed chemical reaction that alters the indicator's color or pattern when it experiences the environmental conditions for which it was intended. Because the response is visual and rapid, chemical indicators are useful for routine checks at the point of use.
Biological indicators work by exposing a known resistant biological challenge to the sterilization process and then incubating the challenge to determine whether any viable organisms remain. The outcome requires time for incubation but provides direct evidence about microbial survival under the conditions applied. This difference in mechanism makes biological indicators a validation tool: they confirm effective microbial inactivation under a specific test scenario.
| Comparison Aspect | Chemical Indicators | Biological Indicators |
|---|---|---|
| Verification Principle | Chemical reaction to heat, steam, and time | Biological response of resistant microorganisms |
| Result Time | Immediate visual color change | Requires incubation period |
| Primary Function | Process exposure confirmation | Microbial inactivation validation |
| Use Frequency | Daily / Every load | Periodic / Scheduled validation |
Typical roles in a sterilization program
Chemical indicators function as process indicators. They are commonly placed inside packages or applied to the outside of wraps to signal that those items passed through a sterilization cycle. Their primary role is immediate operational verification — a quick, visible signal that the package has been exposed to steam.
Biological indicators serve as periodic performance checks for the sterilizer itself. They are used according to facility schedules and regulatory expectations to verify that the sterilizer can achieve microbial inactivation. Because biological testing involves incubation and interpretation, it is not suited for the immediate decision to use an instrument set, but it is important for long-term assurance and validation activities.
Practical application scenarios
In busy central processing departments, chemical indicators are used on each tray and package to mark processed loads during turnover. They support rapid sorting and reduce the risk that unprocessed items are mistaken for processed ones during high-volume handling. In outpatient clinics and dental operatories, small internal indicators provide clinicians with a quick confirmation when assembling sets for patient care.
Biological indicators are typically reserved for scheduled checks, installation qualification, performance qualification, or after maintenance that might affect sterilizer function. They are also used when a facility needs to demonstrate the efficacy of sterilization to an auditor or when validating a new sterilization cycle or load configuration. The incubation period means results are not immediate, so these indicators complement but do not replace the daily visual checks that chemical indicators provide.
Chemical indicators and biological indicators answer different questions about sterilization. Chemical products provide immediate, practical signals that a package experienced sterilization conditions, aiding daily decision making and workflow efficiency. Biological indicators provide evidence of microbial inactivation and serve as a central component of validation and periodic performance checks. Combining these tools within a structured quality program, supported by training, documentation, and routine equipment maintenance, enables healthcare and laboratory teams to manage sterilization processes in a way that aligns operational needs and compliance expectations.

 English
English Français
Français Deutsch
Deutsch Nederlands
Nederlands












 ‘s-Gravenweg 542, 3065SG RotterdamThe Netherlands
‘s-Gravenweg 542, 3065SG RotterdamThe Netherlands
 +31 (0)10 254 28 08
+31 (0)10 254 28 08







