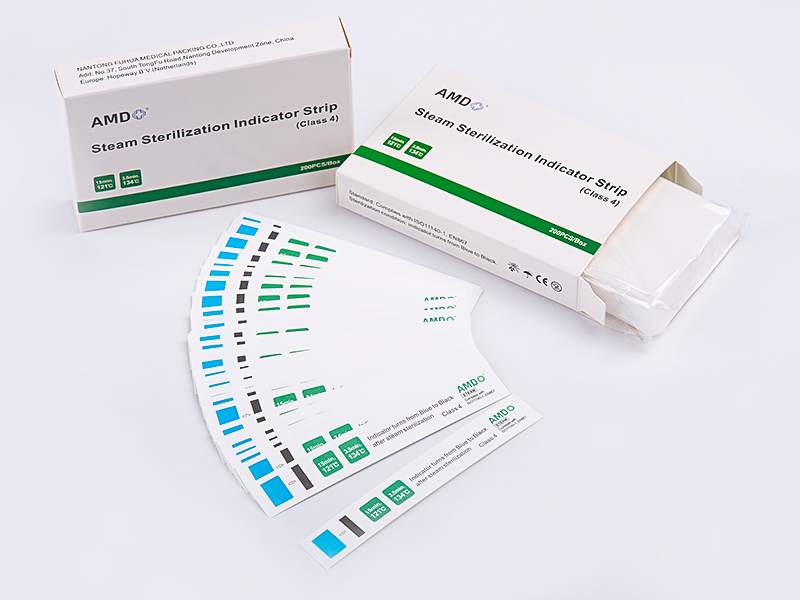
A sterilization solution for routine instrument management should be straightforward, predictable, and compatible with established sterilization processes. The product referenced above is a medical packaging item designed to support the handling, processing, and storage of reusable instruments and small device sets. It brings together a porous paper face and a transparent film backing in a laminated construction, with an integrated closure that does not require separate heat sealing. The result is a single-use package intended to maintain the sterile condition of contents after appropriate processing.
Content
- 1 Design and basic concept
- 2 Intended uses
- 3 Where it is used
- 4 Antimicrobial material: surface protection with a considered approach
- 5 Self-sealing design: convenience, traceability, and controlled access
- 6 Medical-grade paper plus composite film: combining breathability and visibility
- 7 Compatibility with high-temperature, high-pressure sterilization
- 8 Antimicrobial performance: surface treatment versus baseline materials
- 9 Sealing method: integrated closure versus external sealing
- 10 Sterilization safety: material permeability and cycle compatibility
Design and basic concept
The pouch combines a breathable side and a viewing side. The breathable side permits penetration of sterilizing media during processing while providing a microbial barrier after the cycle is complete. The viewing side allows visual verification of the instruments inside without opening the package. A self-closing strip adheres when pressed, creating a seal that is intended for single use. Many versions include an area for process indicators or labeling so personnel can record package contents and processing details. Construction choices focus on allowing validated sterilization methods to reach the instruments while offering a convenient means to store and transport sterile items within a clinical or laboratory setting.
Intended uses
The primary purpose of this packaging is to hold instruments and small assemblies during sterilization and to preserve their sterile status until the moment of use. Typical items placed inside include hand instruments, small handpieces, burs, probes, and other non-porous or wrapped small devices that fit the pouch dimensions. Because the package enables visibility, staff can confirm the correct instruments are present before and after processing without compromising the sealed barrier.
Common sterilization processes employed with this style of package include saturated steam cycles and compatibility with low-temperature methods where indicated by the manufacturer's instructions for use. The pouch is intended to be handled according to standard operating procedures: place cleaned and dried instruments inside without sharp overload, remove excess air, engage the closure, apply any process indicator materials if required, and follow the facility's validated sterilization cycle. After completion and cooling, sealed packages may be stored under recommended conditions until use.
Where it is used
Healthcare facilities of multiple types incorporate this style of packaging into their instrument processing workflows. Hospitals may use pouches for small surgical sets or to manage instruments used in outpatient procedures. Dental practices frequently employ them for routine instrument turnover between patients, taking advantage of the visibility and convenience the pouch provides. Clinical laboratories that perform minor procedural work or that need to maintain sterile instruments for bench use may also adopt similar packaging for specific workflows. In aesthetic and cosmetic procedure settings, practitioners often select disposable packaging that integrates easily with existing sterilization equipment to keep instrument handling efficient and traceable.
Packaging used in instrument processing must balance several demands: it should allow sterilizing agents to reach the content, provide a reliable barrier after processing, enable easy identification, and integrate smoothly into daily workflows.
Antimicrobial material: surface protection with a considered approach
One notable element of the pouch is an antimicrobial treatment incorporated into one or more layers of the material. This antimicrobial component is intended to reduce surface contamination on the package exterior and to limit microbial transfer during handling. The treatment is integrated during manufacture so that the antimicrobial property is continuous across the treated surface rather than applied as a removable coating. The presence of such a material does not replace required sterilization processes, but it does offer an additional layer of precaution for handling and storage prior to use.
Self-sealing design: convenience, traceability, and controlled access
The integrated closure system eliminates the need for separate heat sealing or additional hardware. The pouch features a pressure-activated closure strip that is simple to engage and offers a clear visual cue that the package has been closed. Because the closure is single-use, it provides a straightforward means to indicate whether a package has been previously opened; this can support adherence to instrument use protocols and reduce the risk that a package erroneously reenters sterile circulation.
Medical-grade paper plus composite film: combining breathability and visibility
The pouch construction typically couples a porous paper face with a transparent composite film on the reverse. The paper side is selected for controlled permeability: it allows sterilizing media such as saturated steam to penetrate during processing while serving as a microbial barrier after the cycle. The composite film provides a clear window to view the packaged content, enabling verification without opening the package, and contributes to mechanical strength during transport and storage.
This two-sided construction supports a predictable sterilization outcome while facilitating quick visual checks of instrument identity, orientation, and labeling. The composite film also accommodates labeling and process indicators that rely on visual contrast. Together, these materials enable a balanced approach to barrier function, instrument visibility, and handling robustness.
Compatibility with high-temperature, high-pressure sterilization
The pouch is designed for use with saturated steam sterilization under conditions commonly used for processing reusable instruments, as indicated in the product's instructions for use. Its materials and closure are selected and manufactured to withstand the temperature, pressure, and moisture exposure inherent in such cycles, allowing sterilant access during the active phase and maintaining barrier properties after cooling. When adopting this packaging, users should integrate it into validated sterilization protocols and follow the sterilizer manufacturer's guidance to confirm cycle compatibility.
When selecting packaging for instrument processing, procurement teams and clinical staff frequently weigh practical differences rather than marketing claims.
Antimicrobial performance: surface treatment versus baseline materials
A key difference rests in the treatment applied to the external surface. The branded pouch incorporates an antimicrobial component intended to reduce the level of viable organisms on the package exterior during routine handling. This feature is applied during manufacture so it is integral to the material rather than an after-the-fact coating that can wear away quickly. In contrast, many conventional pouches rely on untreated paper and film surfaces that do not provide active reduction of surface contamination.
Sealing method: integrated closure versus external sealing
Another substantive distinction involves how the package is closed. The branded design uses an integrated self-sealing mechanism that engages through applied pressure along a closure strip. This approach removes the need for separate heat sealing equipment or additional tapes and clips, offering a more compact workflow for areas where heat sealers are not convenient or where rapid turnaround is common. The closure also provides a visual cue that the package has been engaged, which helps staff confirm that a package has been prepared for sterilization.
Conventional pouches commonly depend on a heat-sealable edge or separate sealing processes that require equipment or additional steps. This can introduce more complexity into instrument preparation, especially in smaller clinics or satellite units with limited space for dedicated packaging tools. While heat sealing remains an accepted and valid sealing technique, pressure-activated closures shorten the number of steps and reduce the chance of incorrect sealing technique when used according to the manufacturer's instructions.
Sterilization safety: material permeability and cycle compatibility
Sterilization safety hinges on whether sterilant can reliably reach internal surfaces during the active phase and whether the package maintains a barrier after cycle completion. The branded pouch uses a combination of a porous medical-grade paper face and a composite film back to balance sterilant penetration and postcycle protection. The paper side is selected for controlled permeability so that the sterilizing medium can access the contents, while the film side provides a clear view of the instruments and structural support during handling.
Conventional pouches also pair porous paper with clear film, but variations among base materials, paper porosity. It is therefore important that any packaging option be incorporated into a validated sterilization process and that the facility confirm compatibility with its sterilizer types and cycles. The branded pouch is intended to function with common saturated steam processes when used as instructed, but users should follow sterilization equipment guidance and institutional validation practices to ensure predictable outcomes.
| Feature | Self-Sealing Pouch | Conventional Pouch |
|---|---|---|
| Sealing Method | Integrated pressure closure | Heat sealing required |
| Equipment Requirement | No additional equipment | Requires heat sealer |
| Workflow Efficiency | Faster packaging process | More preparation steps |
| Seal Verification | Visible closure strip | Depends on sealing quality |
The differences between the branded self-sealing pouch and conventional sterilization pouches can be summarized by looking at four areas. Antimicrobial-treated surfaces add a handling-oriented protection layer but do not replace cleaning or sterilization. Integrated pressure-activated closures simplify sealing workflows in environments where heat sealing is less practical. Material combinations and construction influence sterilant access and postcycle barrier properties; compatibility with validated sterilization practices is essential. Storage outcomes depend on proper sealing, correct sizing, and adherence to recommended environmental conditions.
Choosing the appropriate pouch involves matching product features to clinical needs, equipment compatibility, and staff competencies. Thoughtful evaluation based on these operational dimensions will help teams select packaging that fits their processes and supports consistent instrument handling and traceability.

 English
English Français
Français Deutsch
Deutsch Nederlands
Nederlands












 ‘s-Gravenweg 542, 3065SG RotterdamThe Netherlands
‘s-Gravenweg 542, 3065SG RotterdamThe Netherlands
 +31 (0)10 254 28 08
+31 (0)10 254 28 08







